
Label Parts of an Atom — Learning in Hand with Tony Vincent Teaching chemistry, Chemistry
Single-Index Models are high-dimensional regression problems with planted structure, whereby labels depend on an unknown one-dimensional projection of the input via a generic, non-linear, and potentially non-deterministic transformation. As such, they encompass a broad class of statistical inference tasks, and provide a rich template to study statistical and computational trade-offs in the.

Amino Acid Classification and Structure ( Read ) Chemistry CK12 Foundation
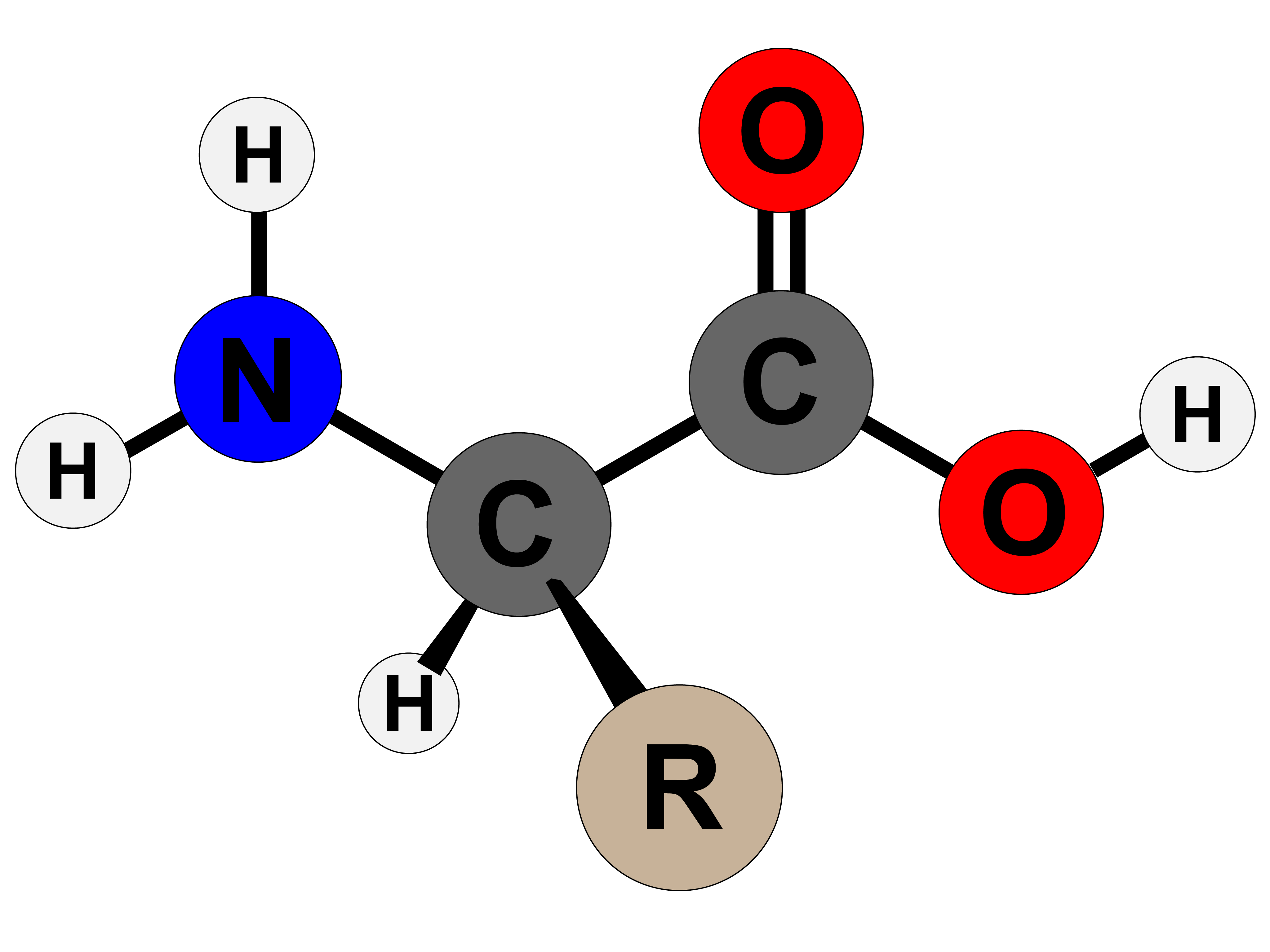
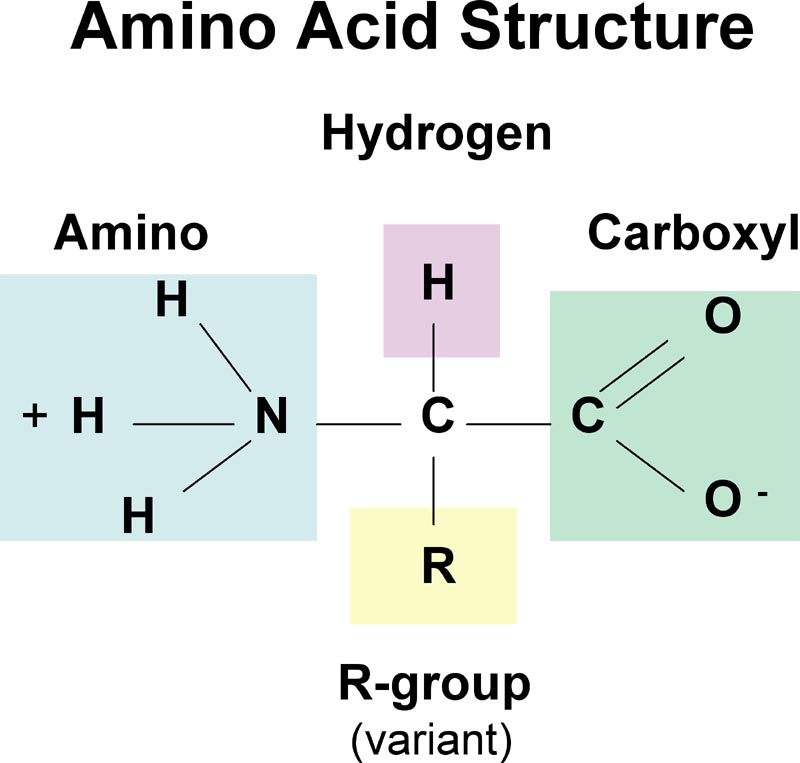
AboutTranscript. Amino acids are the building blocks of proteins. They contain an amino group, carboxylic acid group, alpha carbon, and side chain. Most amino acids have a chiral carbon, which allows them to rotate polarized light. Amino acids can have either an L- or D- configuration, but only the L- form is found in the human body.
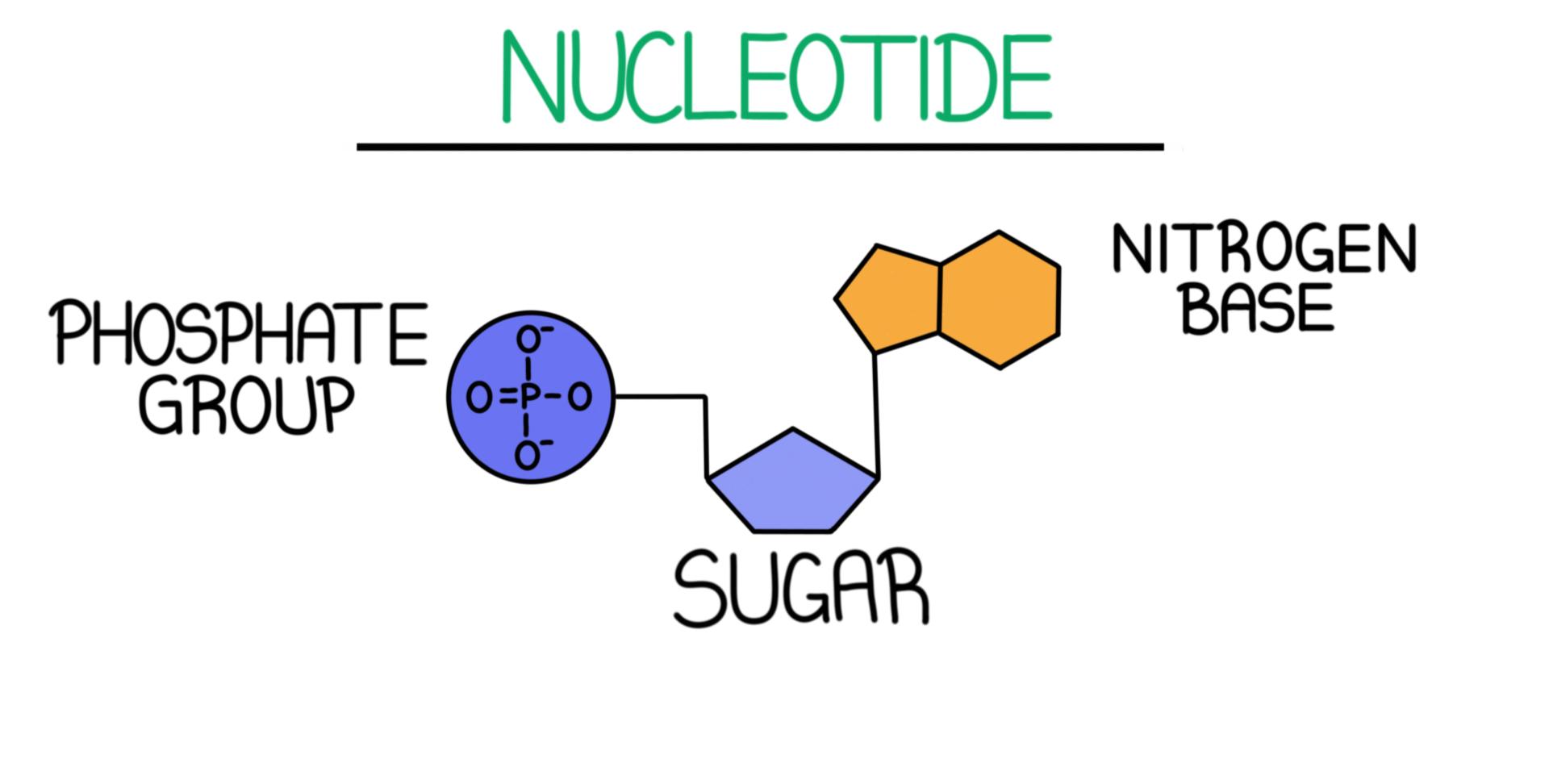
DNA Structure — Overview & Diagrams Expii
More important than identifying features of generic structure components is observing interactions between different features of conceptualisations. This relates to the difference between 'consistency' and 'coherency', which Lakoff and Johnson ( 1980) emphasised. Just as seemingly inconsistent multiple metaphors actually work together.
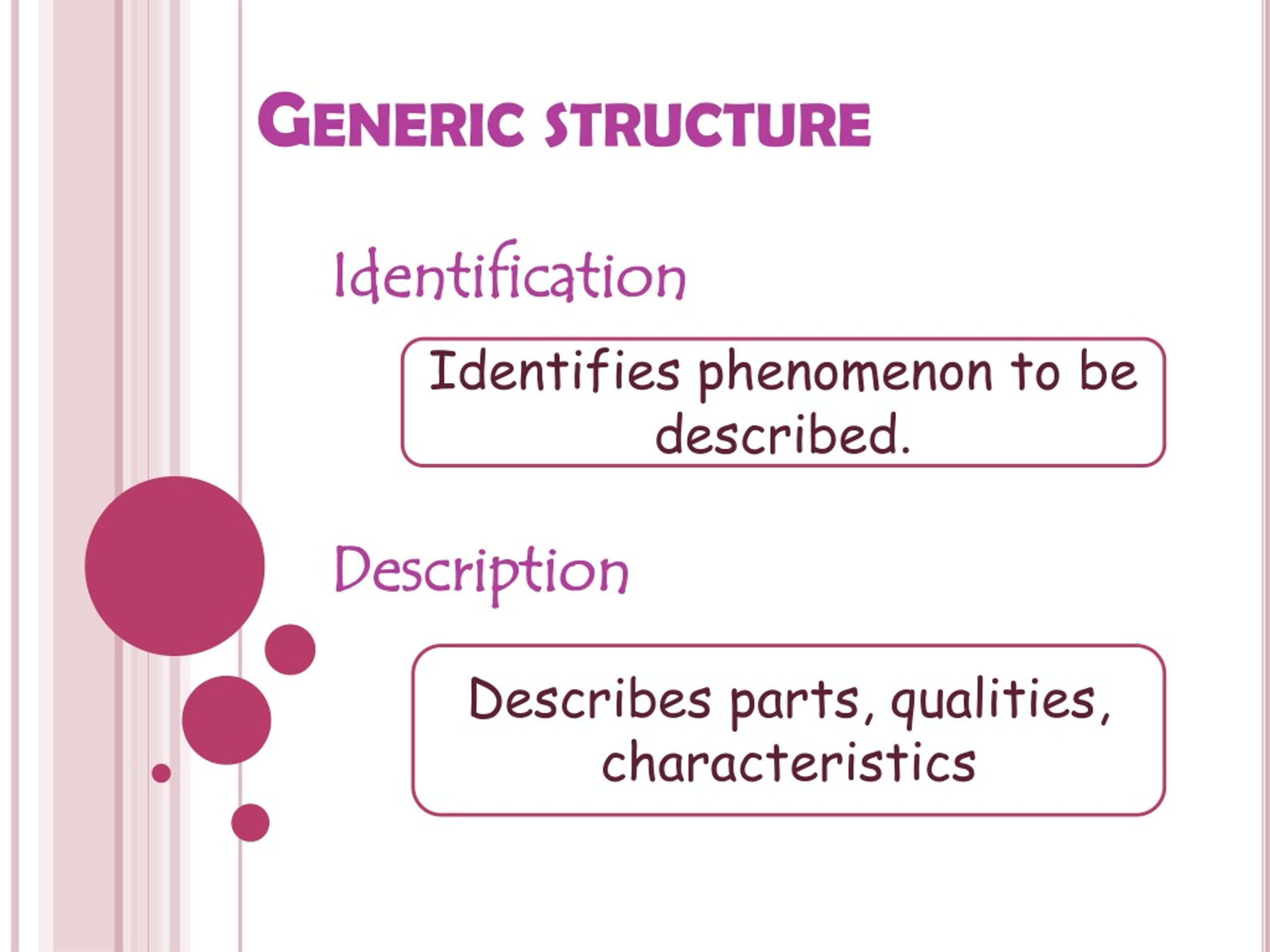
PPT Descriptive Text PowerPoint Presentation, free download ID1036303
The structure of the label and the process of updating safety information affects the evaluation of that information. A comparison of the structure of the EU SmPC and the US PI demonstrates the complexity involved in uniting information derived from two major markets (see Table Table1). 1). Some sections directly lead to another, while other.

PPT PROCEDURE TEXT PowerPoint Presentation, free download ID2501348
August 15, 2021. Pesticide labels are legal documents containing directions for use, handling, storage, and disposal of a pesticide product. With so many sections on a label, it can be difficult to discern what you need to know. This document can help with understanding how a label is designed and what information is provided within each section.
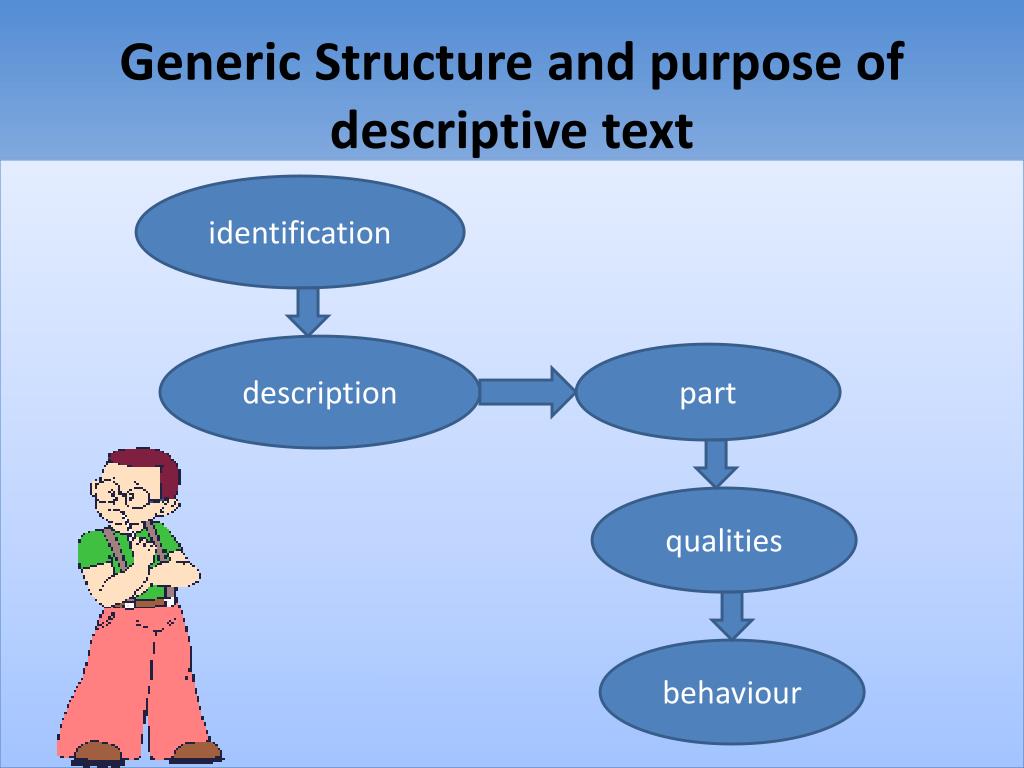
Generic Structure Descriptive Text Berbagi Informasi
Generic Drug-Specific Labeling Guidances and MAPP. Acceptability of Draft Labeling to Support ANDA Approval ( final guidance) Determining Whether to Submit an ANDA or a 505 (b) (2) Application.

Generic Structure Of Descriptive Text
The amino group in beta-alanine has moved to the end of the three-carbon chain. It is a component of pantothenic acid, HOCH 2 C (CH 3) 2 CH (OH)CONHCH 2 CH 2 CO 2 H, a member of the vitamin B complex and an essential nutrient. Acetyl coenzyme A is a pyrophosphorylated derivative of a pantothenic acid amide.
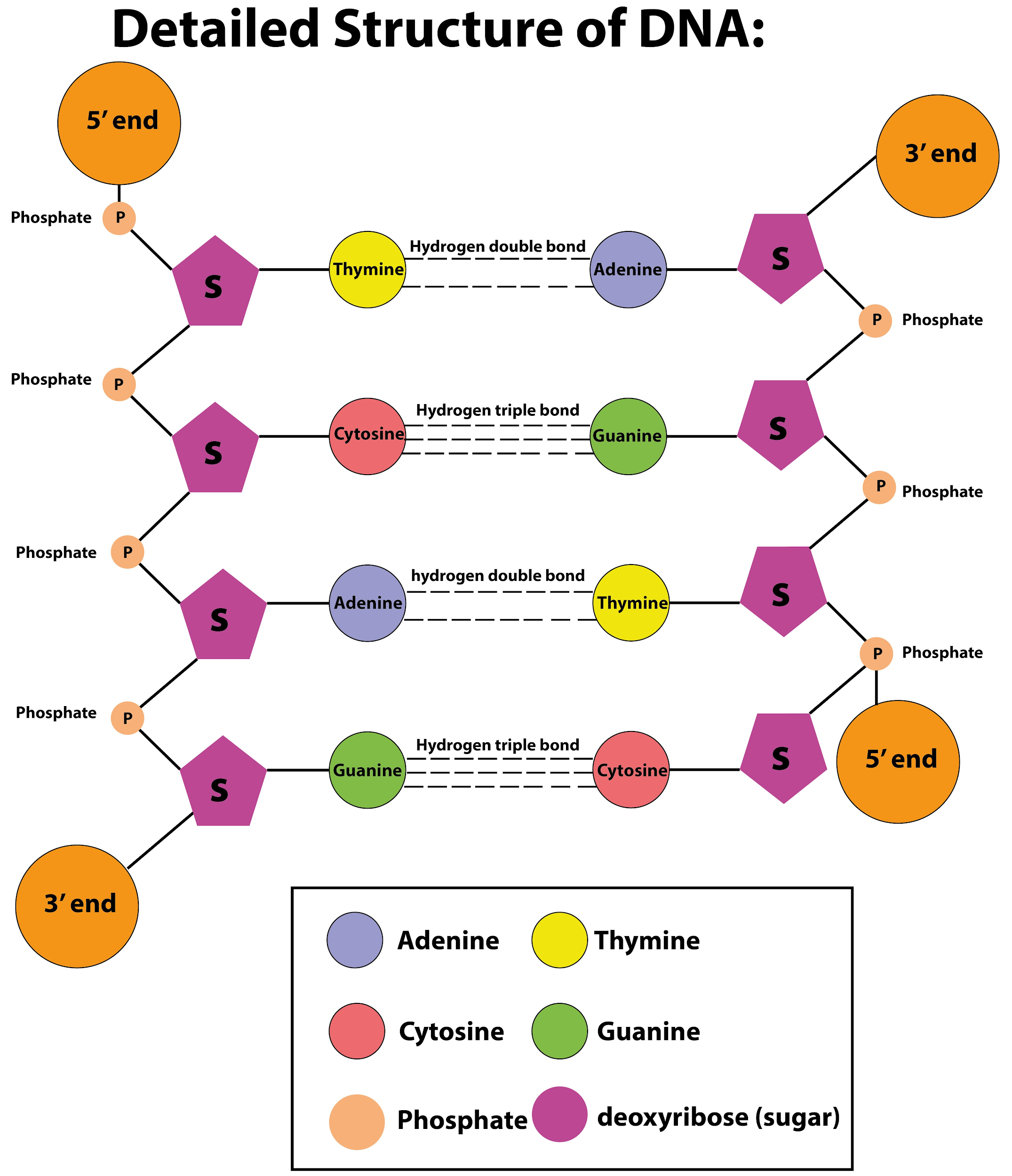
What is the peculiarity of the DNA structure?
Label text memiliki beberapa fungsi, di antaranya. Memberikan informasi rinci tentang suatu produk kepada konsumen. Agar konsumen dapat memilih produk yang baik, aman, sehat dan halal. Agar konsumen terhindar dari efek samping yang membahayakan dari suatu produk. Baca juga: Report Text: Cara Menulis Teks Laporan dalam Bahasa Inggris.

Sketch And Label The Basic Structure Of An Antibody Free Nude Porn Photos
Amino Acid Structure. Amino acids are the monomers that make up proteins. Each amino acid has the same core structure, which consists of a central carbon atom, also known as the alpha (α) carbon, bonded to an amino group (NH2), a carboxyl group (COOH), and a hydrogen atom. Every amino acid also has another atom or group of atoms bonded to the.

Generic structure of the ligands abbreviations (a) L n (n = 13) with... Download Scientific
A Label Contains/Generic Structure of Labels 1. Name of the product, 2. Name of the brand, 3. Description of the product, 4. The ingredients/the contents, 5. The nutrition facts, 6. Suggested uses/doses, 7. Storage, and 8. Expiry date. Language Features of Labels 1. Terms used in the product: serving size, servings per container, dosage, values. 2.

U.S. FDA Food, Beverage and Dietary Supplement Labeling Requirements Viva FDA U.S. FDA
Labeling and Label Approval. FSIS develops and provides labeling guidance, policies and inspection methods and administers programs to protect consumers from misbranded and economically adulterated meat, poultry, and egg products which ensure that all labels are truthful and not misleading. FSIS revised guidance on food product date labeling to.

Amino acid general structure. Download Scientific Diagram
When a drug is approved by the Food and Drug Administration (FDA—the U.S. government agency responsible for ensuring that drugs marketed in the United States are safe and effective), it is given a. Generic (official) name. Brand (proprietary, trademark, or trade) name. For example, phenytoin is the generic name and Dilantin is a brand name.

Report Text And Generic Structure
The typical form has following five components: Structure. Order of fields and logical connections between individual fields. Input fields. Different types of input fields including text fields, password fields, check boxes, radio buttons, sliders. Field labels. Labels describe the meaning of input fields.
Amino_Acid_Structure
Generic (official) name. Brand (proprietary, trademark, or trade) name. For example, phenytoin is the generic name and Dilantin is a brand name for the same drug, which is a commonly used antiseizure drug. The generic name is assigned, in the United States, by an official body—the United States Adopted Names (USAN) Council.
Generic Structure Of Amino Acid
A generic drug is a medication created to be the same as an already marketed brand-name drug in dosage form, safety, strength, route of administration, quality, performance characteristics, and.

Contoh Narrative Text Beserta Generic Structure Dan Language Features
Generic Label Approval. This guidance provides information about procedures for obtaining prior approval for sketch and generic labels for meat and poultry products. It applies to food manufacturers and official establishments seeking to request label consideration and the actions they may take before products may be marketed. It relates to 9.